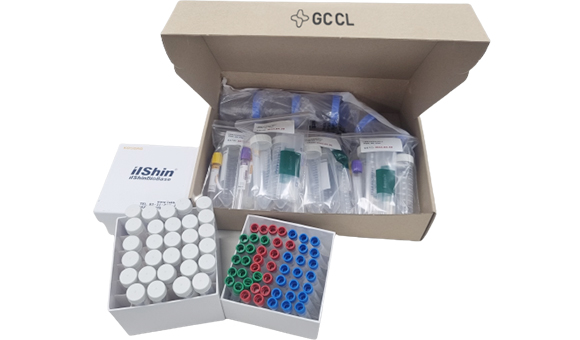
Providing customized lab kit
GCCL creates and provides lab kits optimized for clinical trial protocols.
- Providing lab kit components according to the analysis items by visit date
- Managing the expiration date of lab kits
- On-site training about sample collection and precautions